Fluorescence In Vivo Imaging System
15,000 - 50,000 USD
FOBI is a fluorescence In Vivo imaging system.
There are two main types of optical In Vivo imaging systems: luminescence and fluorescence.
Firefly luciferase and luciferin are mainly used in Luminescence experiments to label cells and obtain images. The light generated by infected luciferase gene is too weak to see with the naked eye. When this is applied to cells and living animals, greater light loss occurs. To capture images of light with such low brightness, imaging system needs to employ an ultra-high sensitivity image sensor.
In the case of fluorescence, either a fluorescent gene or a fluorescent reagent can be used. Both produce light strong enough to be seen with the naked eye. Due to the nature of fluorescence, there is excitation light and emission light, and images can be obtained and analyzed using the emission light from the fluorescent material. At this time, the excitation light is filtered out using a optical filter, and only the emission light required for imaging is passed through. FOBI can effectively obtain excellent In Vivo images by using filters optimized for In Vivo images.
Because luminescent In Vivo imaging devices and fluorescent In Vivo imaging devices have similar yet different principles, key elements such as image sensors and optical filters required for each require different specifications. Therefore, when luminescence and fluorescence are combined into a single device, the product becomes complex and large.
NeoScience manufactures light emitting devices and fluorescent devices separately and faithfully fulfills the functions of each product. And this independent devices simplifies the structure of the product and makes it compact and easy to use and maintain.
FOBI, a fluorescence In Vivo imaging device, can detect most fluorescent substances from blue to NIR and has fast image processing speed. FOBI uses the Defocusing Free HYPER APO lens to capture images for each channel without the need to adjust focus, resulting in clearer image data.

Color Sensor
Fluorescent substances have different colors depending on their wavelength. Imaging devices can capture and analyze images by differentiating between background and fluorescent signals, leveraging the distinctive characteristics of difference color
wavelengths. FOBI uses a color sensor rather than a black-
and-white sensor to suit the characteristics of these fluorescent substances.
When it comes to In Vivo images, unlike cases with little or no reflected light and auto-fluorescence, such as in fluorescence microscopy images, a significant background noise issue is encountered. Generally, cells are transparent and thin, whereas experimental animals or plants used in experiments often have colored or opaque surfaces. Consequently, background noise caused by reflected light or auto-fluorescence can occur, serving as an interfering factor in image analysis. If the camera's sensor is black and white, distinguishing between the signal and the background may become challenging, potentially leading to both being mistakenly identified as signals. However, when a color sensor is used, it becomes possible to differentiate between the signal and the background based on color, resulting in the creation of highly intuitive image data. Thus, the position and size of the signal can be readily identified without the need for any additional image processing.

Easy Imaging
Fluor i In Vivo, which is faithful to its fluorescence function, has a simple structure. So it is easy to use and
maintain.
And the user-friendly program allows you to use it proficiently without any special training.
In addition, the simple structure not only shortens the experiment time by speeding up the imaging speed, but also helps the researcher check the image signal without missing it through immediate response.
The compact size allows efficient use of laboratory space and makes it easy to move the equipment.
HYPER APO Lens
Light is refracted when it passes from one material to another. And the refractive index varies depending on the wavelength of light. The position where the image is formed may vary depending on the wavelength of the light passing through the
lens, due to this phenomenon. FOBI utilizes Hyper
Apochromat technology and can provide image data with focus adjustments corrected for each channel. Images can be captured at a consistent focal length across all channels, resulting in clearer image data and ensuring more accurate measurement results.


DDS (Drug delivery system)
Recently, in the development of new drugs, not only drug efficacy but also targeting is very important. Recently, there has been a significant focus on ensuring that newly developed drugs are precisely targeted to the intended lesion site, delivering their therapeutic effects exclusively to abnormal cells and tissues without impacting normal ones. Although radioactivity can be used to track location, there are many limitations to using radioisotopes and devices in a typical laboratory.
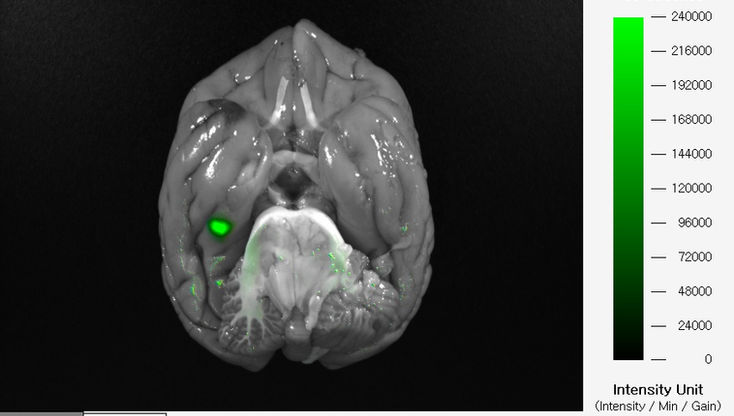
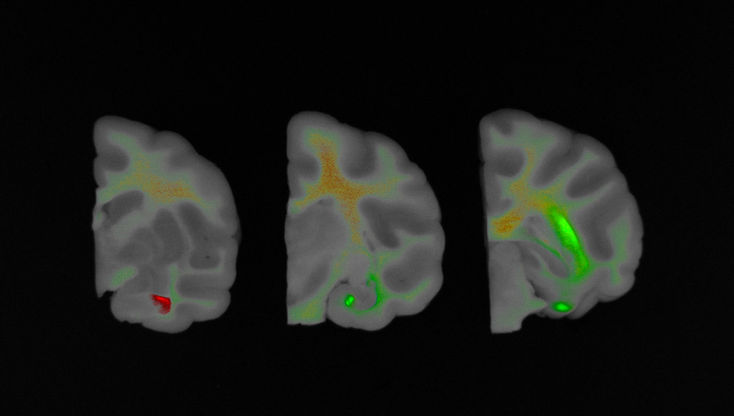
Labeling a newly developed drug with a fluorescent substance and injecting it into a laboratory animal allows you to obtain image data and track its movement using FOBI.
Due to optical limitations, obtaining images of tissues deep inside the animal may not always be possible. In such cases, tracking can be performed using Ex Vivo images. The fluorescence tracked in this way allows for relative quantification by measuring the area and intensity.
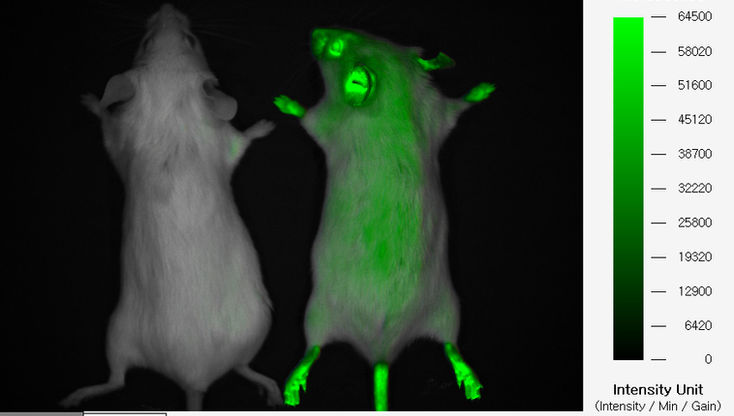
Cell tracking (Tumor, Stem cell)
FOBI is also used for cell tracking. Fluorescent genes like GFP can transfected into tumor cells to make a stable cell line. Stable cell lines can be injected into experimental animals to induce tumor formation, and the tumor size can be measured through imaging. In the case of GFP, it has a short wavelength among fluorescent substances and thus has a short transmittance. Therefore, the use of fluorescent genes such as RFP, mCherry, and iRFP which have longer wavelengths is increasing.
Unlike cancer cells, the characteristics of stem cells or immune cells can change when viruses are used, so fluorescent staining dyes are used more often than fluorescent genes. Since these staining reagents can be cytotoxic when used as is, non-toxic substances with excellent labeling ability are being developed using various nanoparticle-like structures.
FOBI can capture images of labeled cancer cells, stem cells, and immune cells and get quantitative data. You can track the cancer growth process and cell migration path.

Plant Fluorescence Imaging
FOBI can be used to monitor the expression of specific
genes in plants. Without imaging equipment like FOBI,
preparing plant samples into which specific genes have been introduced can be a time-consuming process. After gene transfection, confirming the presence of the gene involves planting seeds, waiting for sprouting, and then using methods like PCR to confirm its existence. Subsequently, when harvested, the plant sample containing the introduced gene is prepared, which can take as long as one plant life cycle.
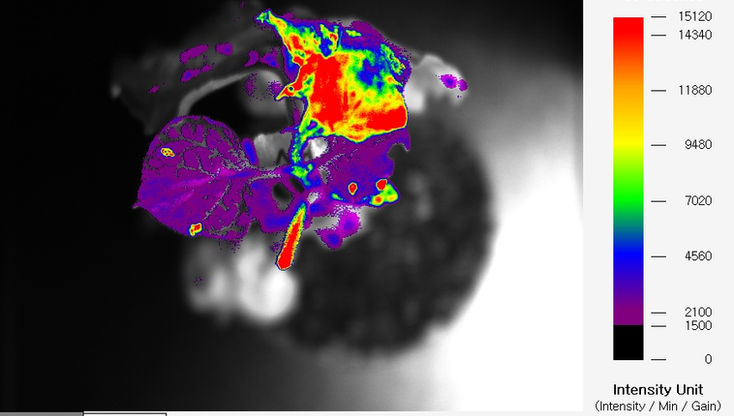
However, by utilizing fluorescent genes, it becomes possible to visually confirm whether a gene has been introduced, and only the introduced seeds can be promptly selected and tested. Moreover, it is possible to verify gene expression not only in plant seeds but also in specific parts of leaves or stems. Obtaining fluorescence images like GFP in plant leaves can be challenging due to the strong autofluorescence of chlorophyll.
FOBI can remove the interference caused by chlorophyll's
autofluorescence through optimized filters. This allows for the separation of only the GFP signal, providing clear image and quantitative data.

Other Fluorescence Imaging
The distinction between FOBI and a fluorescence microscope lies in their magnification functions. This applies to various samples, including drugs, cancer cells, stem cells, and plants. Regardless of the sample
type, FOBI allows for the capture of non-magnified fluorescence images, which in turn enables the
acquisition of high-quality quantitative data.
Furthermore, microorganisms can be labeled with fluorescence and tracked from the mouth, through the intestines, and all the way to the anus. Specific microorganisms can be labeled with fluorescent dye, mixed with contaminated water, and passed through a developed water purification filter to obtain an image. This process helps gauge the effectiveness of microorganism filtration. Additionally, using POCT's
probe as a fluorescent substance, Fluor i In Vivo can provide both
images and quantitative data, even for substances with low concentration. Fluorescence images without magnification currently find diverse applications, and it is anticipated that more experiments and applications will emerge in the future.


Optimized Filter
FOBI employs filters optimized for fluorescence In Vivo imaging. This has different characteristics from fluorescence microscopy. When viewing cells under magnification, autofluorescence is rarely a concern. Therefore, by utilizing a filter with a narrow range that closely matches the wavelength of the fluorescent substance, exceptional fluorescence images can be obtained.
NeoScience conducts various filter tests to select the most suitable filter for In Vivo imaging applications. As a result, we can get intuitive, rapid, and clear image data.
ICG (Indocyangreen) Tail Vein Injection
User Friendly Program
FOBI's program is designed user friendly.
You can monitor the live window and data video through the central panel. The fluorescence intensity can be observed using the scale bar, which is adjustable. This allows for a comparison and analysis of fluorescence intensity with other image data. The scale bar can also be customized with different colors, making it easy to intuitively assess fluorescence intensity.
Device control is accessible through the panel on the right. Depending on the signal strength, exposure time and gain can be adjusted to obtain accurate quantitative values. Basic information about image data can be found in the lower-right panel, along with quantitative values.
File and folder management is possible through the left panel, where image data are distinguishable through thumbnails. With a simple double click, the image can be opened in the middle panel. Data can be organized separately by user and experiment. Most functions are represented by icons, making it easy for users to navigate without requiring special training. Each icon is designed intuitively, providing clear insight into its function.

Specifications